 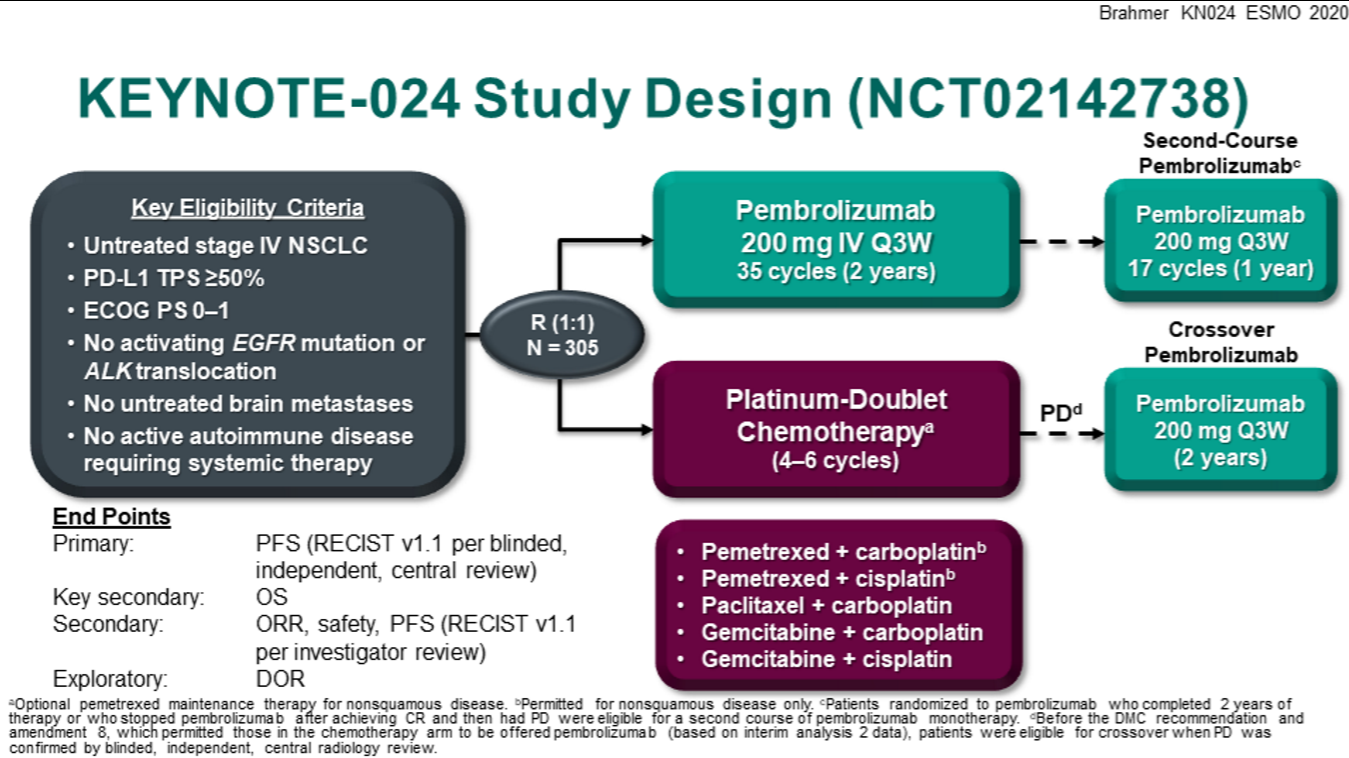
Participants randomized to SOC chemotherapies will be treated with their randomized study drug for up to 4-6 cycles. 
Treatment Phase: Participants randomized to pembrolizumab will be treated for up to 35 cycles or until documented progressive disease (PD) occurs. Condition or diseaseĭrug: Pembrolizumab Drug: Paclitaxel Drug: Carboplatin Drug: Pemetrexed Drug: Cisplatin Drug: Gemcitabine With Amendment 09 (20 December 2017), once participants have achieved the study objective or the study has ended, participants will be discontinued from this study and enrolled in an extension study to continue protocol-defined assessments and treatment. The primary hypothesis of this study is that participants with PD-L1 strong NSCLC will have a longer Progression Free Survival (PFS), as assessed by Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1) when treated with pembrolizumab than when treated with SOC platinum-based chemotherapies. 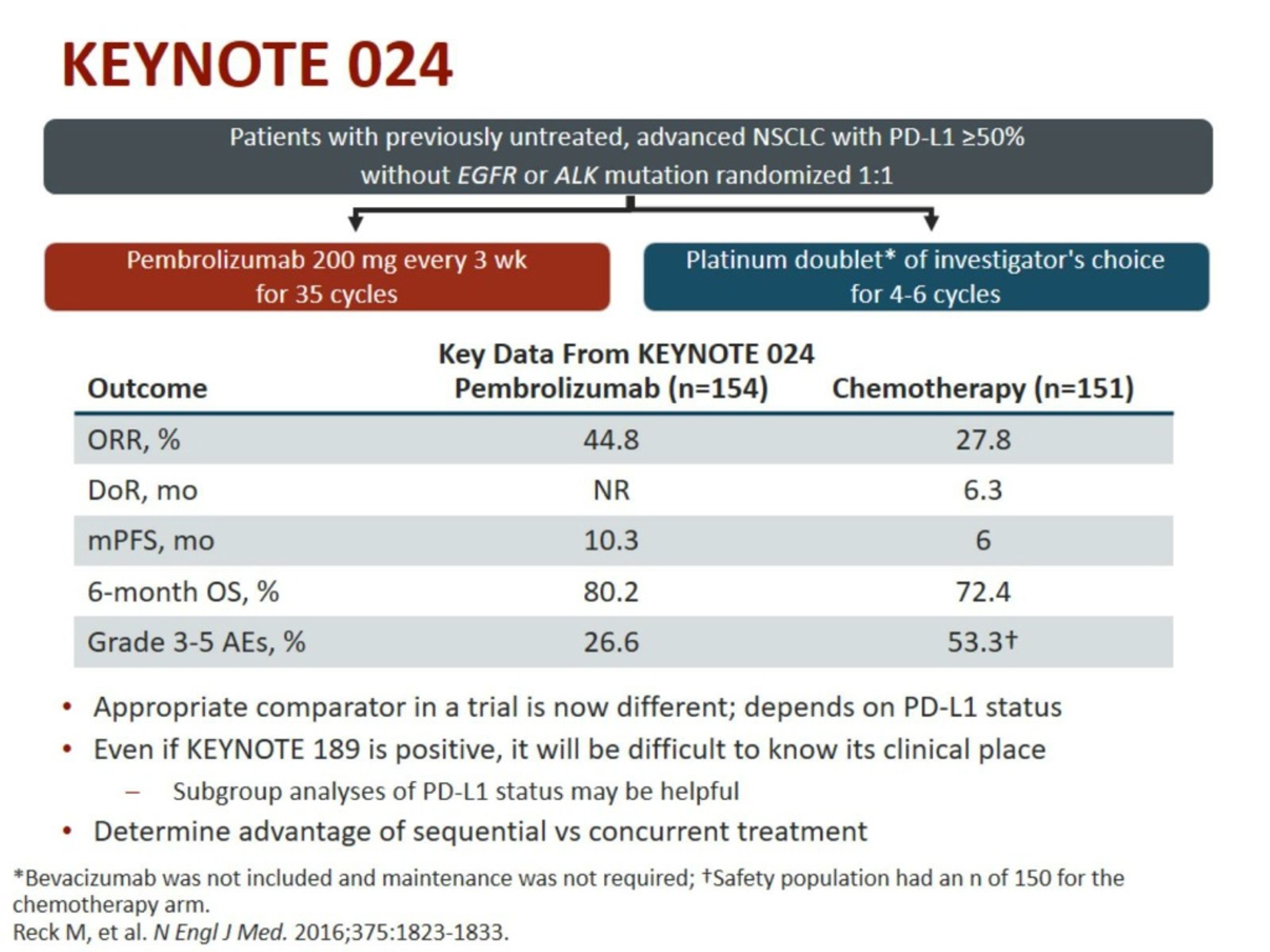
This is a study to assess the efficacy and safety of pembrolizumab (MK-3475/SCH 900475) compared to standard of care (SOC) platinum-based chemotherapies in the treatment of participants with previously untreated stage IV, programmed cell death ligand 1 (PD-L1) strong expressing Non-Small Cell Lung Cancer (NSCLC). Why Should I Register and Submit Results?. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
